Reactive Force Field (ReaxFF)
Understanding ReaxFF
In many molecular dynamics simulations, atoms interact through predefined potentials such as Lennard–Jones or Morse. These models assume that the bonding structure between atoms does not change.
However, many physical and chemical processes involve chemical reactions, where bonds continuously break and form. Examples include combustion, catalysis, battery chemistry etc.
The Reactive Force Field (ReaxFF) was developed to simulate such systems. It allows atoms to dynamically create and destroy bonds during the simulation, making it suitable for modeling realistic chemical processes.
Bond Order Concept
The central idea of ReaxFF is the bond order. Instead of defining fixed bonds between atoms, the strength of a bond depends continuously on the interatomic distance.
A simplified expression for bond order is
\[ BO_{ij} = e^{-p_1 r_{ij}} + e^{-p_2 r_{ij}} + e^{-p_3 r_{ij}} \]Here, \( r_{ij} \) is the distance between atoms \(i\) and \(j\), while \(p_1\), \(p_2\), and \(p_3\) are parameters fitted to quantum chemistry calculations. Since BO has no unit, so unit of p is \(\AA^{-1}\).
When atoms move closer together, the bond order increases, representing bond formation. When atoms move apart, the bond order decreases, eventually leading to bond breaking.
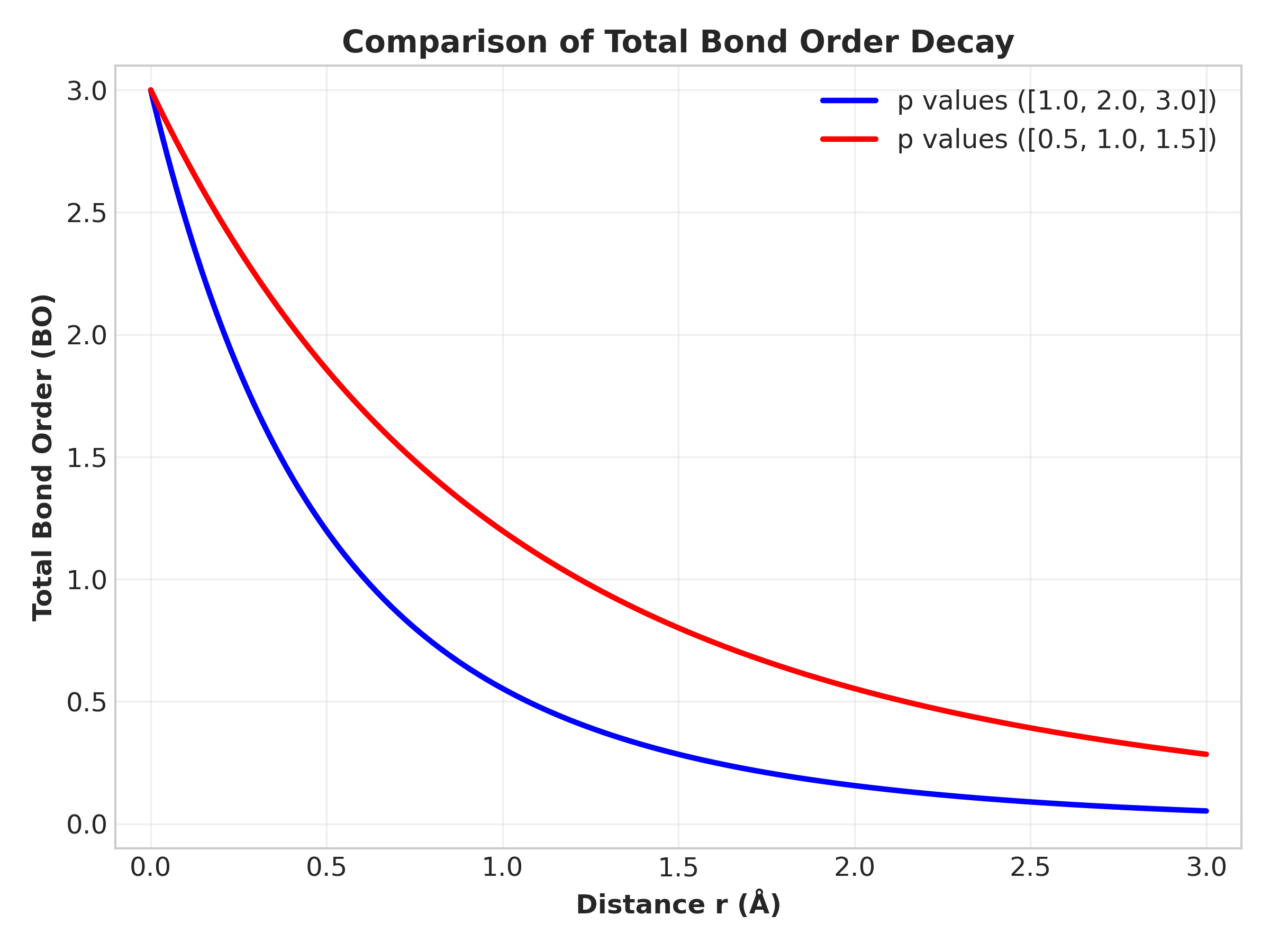
From the graph, we can clearly see that bond formation happens gradually, no sharp peak.
Example implementations and simulation codes can be found at GitHub.